Potential mechanisms of microtubule plus-end tracking. (A) Motor-driven... | Download Scientific Diagram
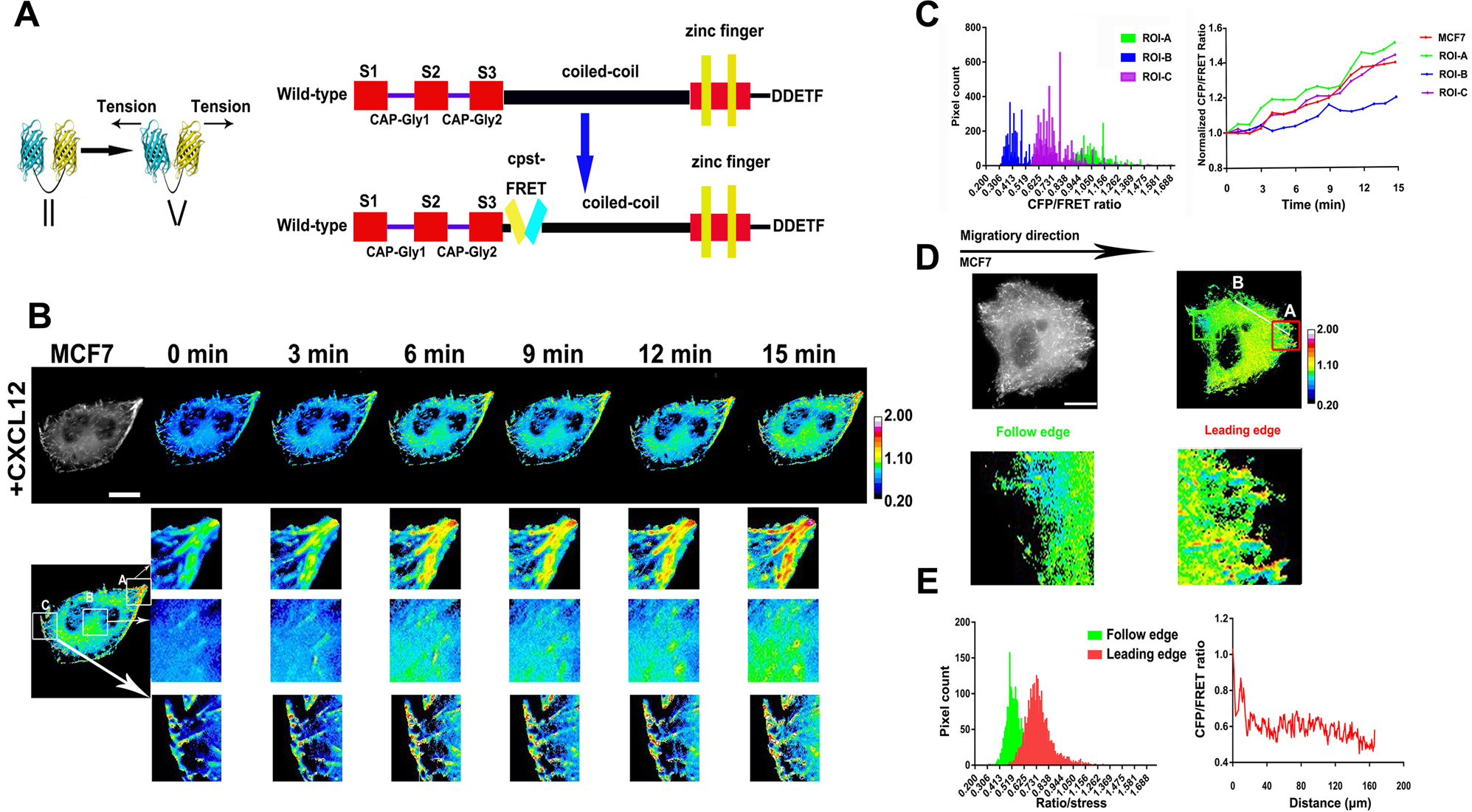
CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engageme

The CLIP-170 N-terminal domain binds directly to both F-actin and microtubules in a mutually exclusive manner - Journal of Biological Chemistry

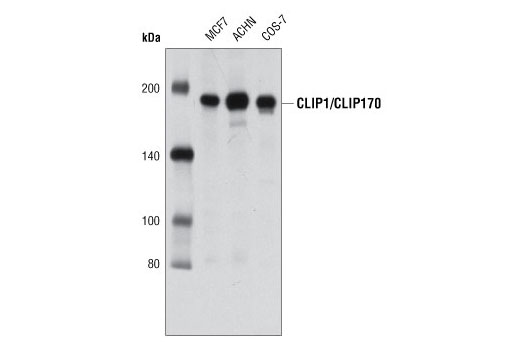
Overexpression of the microtubule-binding protein CLIP-170 induces a +TIP network superstructure consistent with a biomolecular condensate | bioRxiv
Overexpression of the microtubule-binding protein CLIP-170 induces a +TIP network superstructure consistent with a biomolecular condensate | PLOS ONE

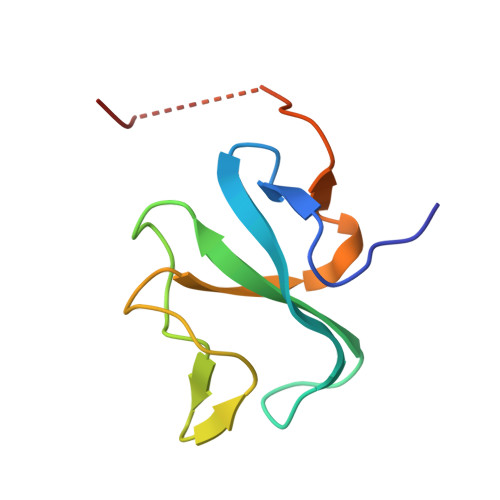
Structural basis for tubulin recognition by cytoplasmic linker protein 170 and its autoinhibition | PNAS
CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engageme

CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engagement - ScienceDirect

The microtubule plus-end-tracking protein CLIP-170 associates with the spermatid manchette and is essential for spermatogenesis

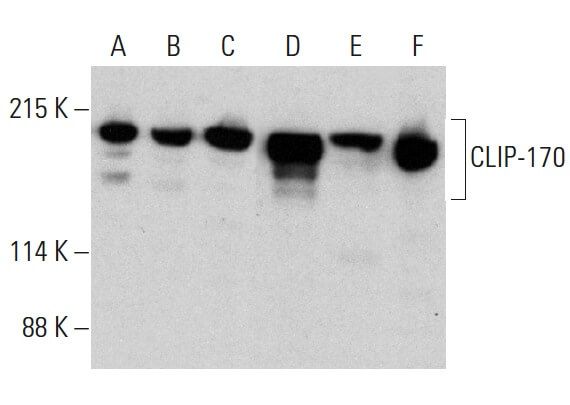
Microtubule binding proteins CLIP-170, EB1, and p150Glued form distinct plus-end complexes - ScienceDirect

Ubiquitination of CLIP-170 family protein restrains polarized growth upon DNA replication stress | Nature Communications
Overexpression of the microtubule-binding protein CLIP-170 induces a +TIP network superstructure consistent with a biomolecular condensate | PLOS ONE

The microtubule plus-end-tracking protein CLIP-170 associates with the spermatid manchette and is essential for spermatogenesis

Ninein is essential for apico-basal microtubule formation and CLIP-170 facilitates its redeployment to non-centrosomal microtubule organizing centres | Open Biology