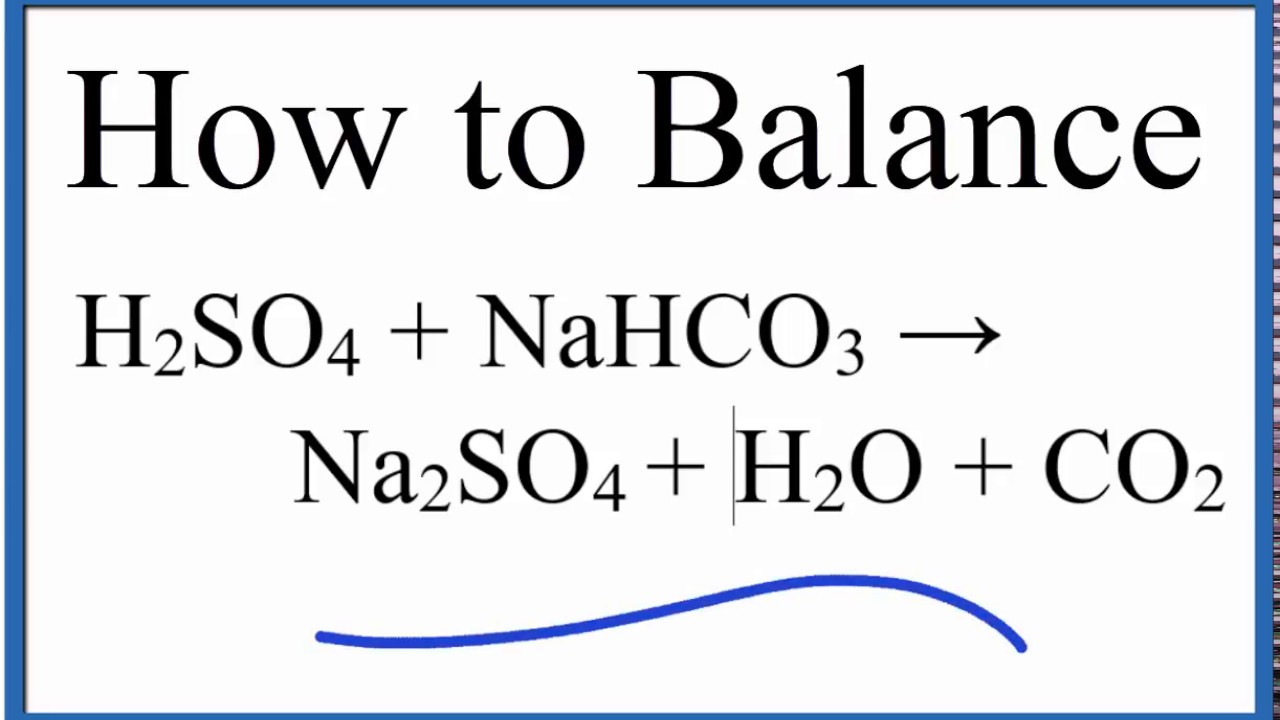
SOLVED: Aqueous solutions of sodium bicarbonate and sulfuric acid react to produce carbon dioxide according to the following equation: 2 NaHCOs(aq) + H,SO4(aq) 3 2CO2(g) + NazSO4(aq) + 2 HzO() If 105.4

Write the balanced ionic equation for the reaction of sodium bicarbonate with sulphuric acid - YouTube

What does a sodaacid type fire extinguisher contain How does it work Explain the working of acid fir...